Review Article
Author: Christos Chapeshis
April 21, 2026
Gerontologist, CEO of ABeWER
BScN, Dipl. W, Dipl. N, Dipl. CN, RN, CDTT, MScG
Abstract
Background
Pressure injuries (PIs) represent a major global healthcare burden, particularly among immobilized, elderly, and critically ill populations. Optimal wound dressing selection is still a central yet unresolved challenge, particularly when stratified by injury stage and wound microenvironment. Despite comprehensive international guidelines, variability persists in clinical decision-making, often driven by uncertainty in differentiating wound characteristics such as exudate level, infection status, and tissue viability.
Objective
To critically evaluate stage-specific wound dressing selection in pressure injuries, integrating current guideline-based evidence with biomechanical and microenvironmental considerations, and to contextualize dressing efficacy within advanced support surface technologies.
Methods
A narrative synthesis was carried out using international guidelines (NPIAP/EPUAP/PPPIA), systematic reviews, randomized controlled trials, and high-quality observational studies. Emphasis was placed on stage-specific interventions, infection management, and the interaction between dressings and mechanical offloading systems.
Results
Dressing selection must be individualized based on wound stage, exudate, infection, and patient-centered risk factors. Evidence supports the use of foam, hydrocolloid, alginate, and antimicrobial dressings in distinct clinical scenarios. However, dressing efficacy is significantly modulated by biomechanical factors, including pressure redistribution, shear reduction, and microclimate control—parameters strongly influenced by support surface technology.
Conclusion
Stage-specific dressing selection cannot be isolated from the mechanical environment of the wound. Integration with advanced support surfaces, particularly dynamic systems such as multifunctional lateral turning mattress systems, constitutes a crucial progress in pressure injury management and limb preservation.
Keywords
Pressure injury; wound dressing selection; stage-specific treatment; heel pressure injury prevention; device-related pressure injury; exudate management; infection control; support surfaces; microclimate; limb preservation
1. Introduction
Pressure injuries arise from sustained mechanical loading, leading to ischemia, tissue deformation, and ultimately necrosis. Their pathophysiology is multifactorial, involving pressure, shear, microclimate, and tissue tolerance (Gefen, 2014). In spite of advances in prevention, prevalence remains high, particularly in long-term care and intensive care settings.
The International Clinical Practice Guideline (NPIAP/EPUAP/PPPIA) provides over 100 recommendations addressing prevention and treatment, yet acknowledges persistent gaps in dressing selection and clinical decision-making (Haesler, 2019). These gaps are reflected in clinician search behavior, which increasingly focuses on high-intent, decision-based queries, such as “what dressing for stage 3 pressure ulcer with slough” or “best dressing for infected pressure ulcer.”
This article intends to bridge the divide between guideline recommendations and bedside decision-making through providing a stage-specific, mechanistically grounded framework for wound dressing selection, integrated with contemporary advances in support surface technology.
2. Methods
A structured narrative review methodology was employed:
- Primary sources: International guidelines (NPIAP/EPUAP/PPPIA 2019 & 2026 updates)
- Secondary sources: Systematic reviews and meta-analyses (Cochrane Library, PubMed)
- Clinical studies: Randomized controlled trials and prospective cohort studies
- Supplementary analysis: Biomechanical and translational wound healing literature
Search strategy included terms such as:
- “stage-specific pressure ulcer dressing.”
- “foam vs hydrocolloid pressure injury.”
- “pressure ulcer infection management.”
Evidence was stratified according to established hierarchies, with guideline-based recommendations prioritized.
3. Results: Stage-Specific Dressing Selection
3.1 Stage 1 Pressure Injury
Clinical Characteristics
- Non-blanchable erythema
- Intact skin
- No exudate
Dressing Strategy
- Prophylactic silicone foam dressings
- Transparent films
Rationale
The main objective is protection and microclimate stabilization, not absorption. Silicone foam dressings reduce friction and shear while preserving optimal hydration.
Evidence
Randomized trials demonstrate that prophylactic foam dressings reduce the incidence of pressure injuries by up to 60% in high-risk patients (Santamaria et al., 2015).
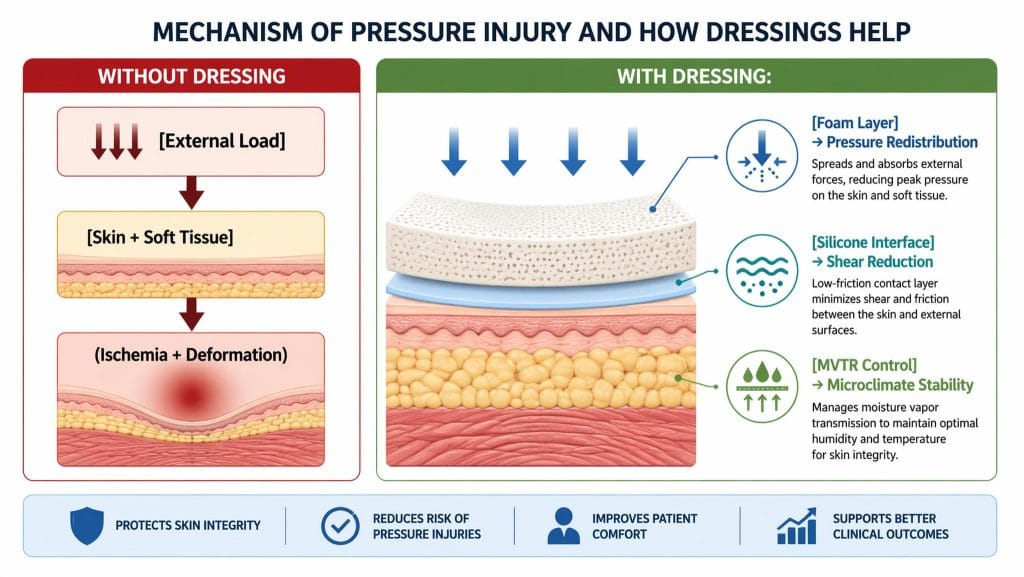
Image 1. Pathophysiological Targets of Prophylactic Dressings
Evidence-Based Dressing Selection (Industry-Specific)
Within Stage 1 pressure injury management, the selection of prophylactic dressings should be guided by biomechanical performance (pressure redistribution, shear attenuation), moisture vapor transmission rate (MVTR), and atraumatic adhesion properties, as emphasized by international guidelines.
Silicone Foam Dressings (High-Level Evidence: RCTs, Systematic Reviews)
- Mölnlycke Health Care (Sweden): Mepilex Border Sacrum/Heel—extensively studied; demonstrates significant reduction in sacral and heel pressure injuries (RR reduction ~50–60%) in ICU populations (Santamaria et al., 2015; Forni et al., 2018).
- Smith+Nephew (UK): Allevyn Life—multilayer foam with hyper-absorptive core and cushioning effect; observational and cohort data imply improved microclimate control and reduced shear-related injury.
- Convatec Group (UK): AQUACEL Foam—hydrofiber interface supports moisture balance; indirect evidence supports prophylactic use in at-risk skin.
- Coloplast (Denmark): Biatain Silicone—demonstrates favorable exudate handling despite Stage 1 being non-exudative, due to its microclimate buffering capacity.
- Paul Hartmann AG (Germany): HydroTac / Zetuvit Silicone Border—widely used in European practice; strong clinician-reported outcomes in pressure redistribution.
- Urgo (France): UrgoTul Border—lipido-colloid interface reduces friction and preserves skin integrity.
Transparent Films (Moderate Evidence)
- Solventum (formerly 3M Health, USA): Tegaderm Film—gold standard for low-profile protection; supports visual inspection while maintaining hydration and reducing external contamination. However, the shear reduction is lower than that of foam.
- Cardinal Health (USA) and Medline Industries (USA): provide cost-effective film dressings; commonly used in large-scale prevention protocols in which monetary considerations are relevant.
Adjunct/Hybrid Technologies
- Integra LifeSciences (USA) and B. Braun (Germany): contribute to advanced wound interfaces and protective dressings, though evidence for pure prophylactic use in Stage 1 remains less robust and often extrapolated from treatment data.
Quantitative Outcomes from Literature
- Relative risk reduction: 40–60% (foam vs. standard care)
- Number needed to treat (NNT): ~8–12 patients to prevent one pressure injury (Santamaria et al., 2015)
- Heel-specific reduction: up to ~50% in high-risk cohorts (Forni et al., 2018)
Importantly, the heterogeneity in dressing composition (thermal insulation, stiffness, adhesion) is still a limitation in comparative effectiveness research, as highlighted by the Prophylactic Dressing Standards Initiative.
Critical Interpretation
While silicone foam dressings have the strongest evidence base, their benefit appears most pronounced in high-risk populations (e.g., immobility, ICU admission, vascular compromise). Transparent films may be appropriate for low-risk Stage 1 presentations, but they lack sufficient biomechanical protection in anatomically vulnerable areas (e.g., the heel and sacrum).
Clinical Implication
Stage 1 pressure injury management ought to prioritize:
- Early identification (per guideline recommendations)
- Immediate offloading
- Selective use of multilayer silicone foam dressings in high-risk anatomical zones
Uncertainty Statement
Despite strong RCT data, uncertainty persists regarding:
- Optimal dressing thickness and stiffness
- Long-term cost-effectiveness across health care systems
- Standardization of prophylactic dressing performance measures
Further high-quality comparative trials and standardized testing frameworks are required.
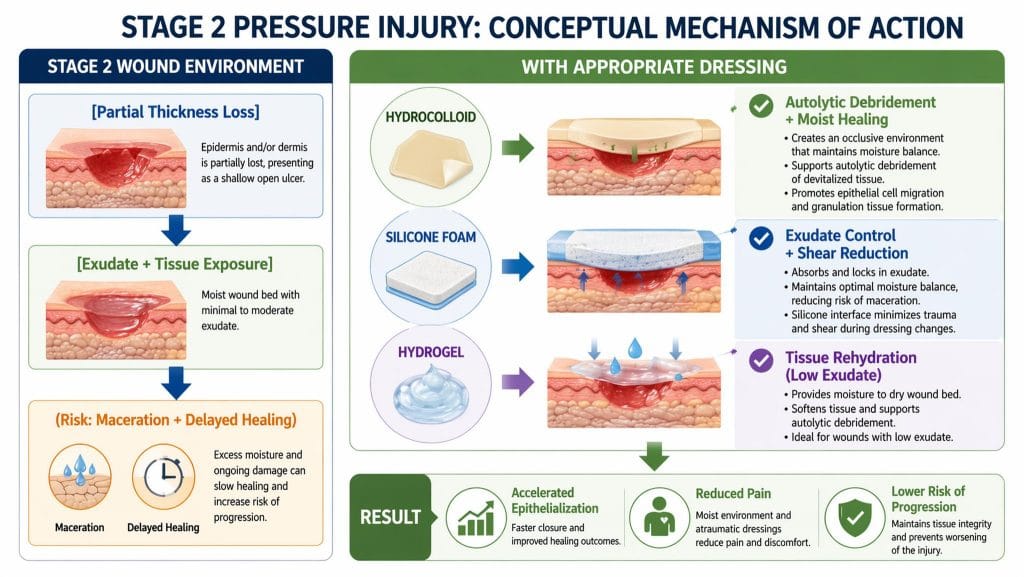
3.2 Stage 2 Pressure Injury
Clinical Characteristics
- Partial-thickness skin loss
- Shallow ulcer
- Minimal to moderate exudate
Dressing Options
- Hydrocolloids
- Silicone foam dressings
- Hydrogels (low exudate)
Clinical Challenge
Differentiation from moisture-associated skin damage (MASD) is critical, as misclassification leads to inappropriate treatment.
Rationale
Hydrocolloids promote autolytic debridement and maintain a moist environment, facilitating epithelialization.
Evidence-Based Dressing Selection and Industry Mapping
In Stage 2 pressure injuries, the therapeutic objective transitions from pure prevention to active epithelial regeneration, while preserving an optimal moist wound environment, as strongly recommended in international guidelines. Unlike Stage 1, exudate management and atraumatic dressing removal become clinically decisive.
Hydrocolloid Dressings (Moderate–High Evidence)
Hydrocolloids remain a keystone for superficial partial-thickness injuries.
- Convatec Group (UK): DuoDERM®—extensively studied; promotes autolytic debridement and accelerates epithelialization. Meta-analyses indicate a ~20–30% reduction in healing time compared with gauze (Heyneman et al., 2008).
- Coloplast (Denmark): Comfeel®—demonstrates strong occlusive properties; effective in low-to-moderate exudate environments.
- B. Braun (Germany): Askina Hydro—widely used in European settings; good conformity to anatomical surfaces.
Silicone Foam Dressings (High-Level Evidence: RCTs, Cohort Studies)
Highly valuable when exudate is moderate or anatomical areas are exposed to shear.
- Mölnlycke Health Care (Sweden): Mepilex Border—atraumatic silicone interface reduces pain and tissue damage; RCTs display improved healing trajectories and reduced dressing-related trauma.
- Smith+Nephew (UK): Allevyn Gentle Border—combines absorption with pressure redistribution; observational studies suggest reduced peri-wound maceration.
- Paul Hartmann AG (Germany): HydroTac Comfort—hydroactive foam with moisture regulation properties.
- Medline Industries (USA) and Cardinal Health (USA): cost-effective silicone foams used in institutional protocols with comparable clinical outcomes when applied appropriately.
Hydrogels (Selective Indication: Low Exudate)
- Integra LifeSciences (USA) and Urgo (France): hydrogels (e.g., Intrasite Gel, Urgo Hydrogel) facilitate rehydration of desiccated tissue and support autolysis; particularly relevant in ischemic or dry Stage 2 lesions.
Adjunct Transparent Films
- Solventum (USA): Tegaderm™ may serve as a secondary dressing or protective cover; however, due to limited absorptive capacity, it is not recommended as a primary dressing in exudative Stage 2 wounds.
Biomechanical and Microenvironmental Considerations
Stage 2 injury progression is driven by superficial tissue deformation and microvascular compromise, compounded by moisture imbalance. Dressings must therefore address three axes:
- Moisture Balance → Hydrocolloids maintain occlusion and support keratinocyte migration
- Shear Reduction → Silicone interfaces decrease mechanical trauma.
- Exudate Control → Foam matrices prevent maceration.
The absence of uniform comparative testing across dressings is still a limitation, as highlighted by the Prophylactic Dressing Standards Initiative.
Image 2. STAGE 2 WOUND ENVIRONMENT
Clinical Outcomes and Statistics
- Hydrocolloids vs. traditional dressings:
→ Healing rate improvement: ~20–40% (systematic reviews) - Silicone foam dressings:
→ Reduced dressing-related trauma by up to 50% (clinical cohort data) - Moist wound healing environments:
→ Epithelial migration increased by 2–3× compared with dry conditions (Winter, 1962; foundational model)
Critical Interpretation
Although hydrocolloids remain highly effective, their use is limited in moderate-to-high exudate wounds due to the risk of maceration. In contrast, silicone foam dressings present a broader safety margin across exudate levels and anatomical sites, particularly in the lower limb, where edema and shear forces are prevalent. Hydrogels should be reserved for carefully selected cases due to limited absorptive capacity.
Clinical Implications
Management of Stage 2 pressure injuries requires:
- Accurate differentiation from MASD
- Selection of dressing based on exudate level and biomechanical risk
- Continuous reassessment to prevent progression to deeper tissue injury
Uncertainty Statement
Despite extensive clinical use, there is heterogeneity in study design, outcome measures, and product composition. Direct one-to-one RCTs comparing modern silicone foams versus hydrocolloids remain limited, and extrapolation across anatomical sites (e.g., sacrum vs. heel) should be undertaken cautiously.
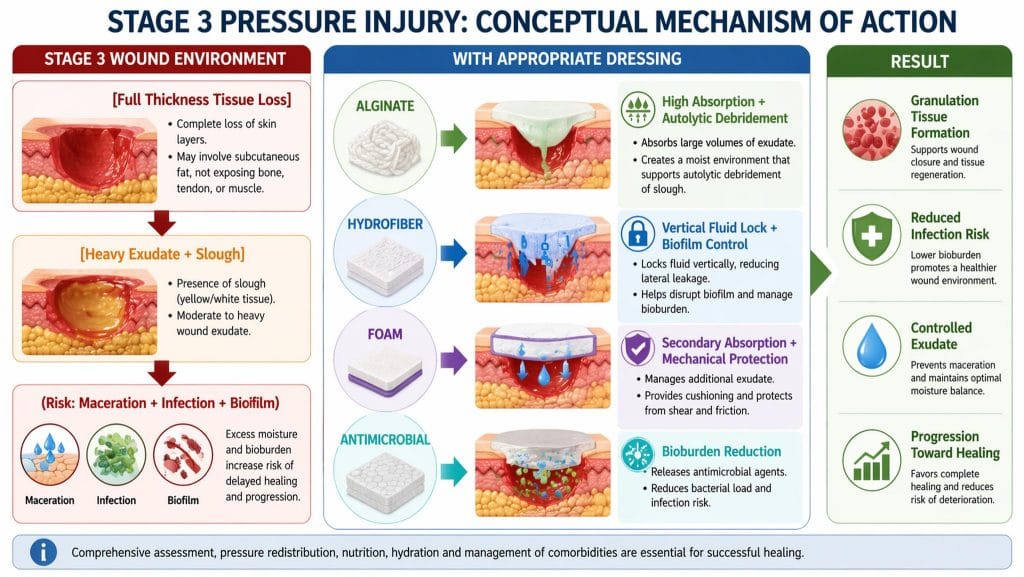
3.3 Stage 3 Pressure Injury
Clinical Characteristics
- Full-thickness skin loss
- Possible slough
- Moderate to heavy exudate
Dressing Options
- Alginate dressings
- Hydrofiber dressings
- Foam dressings
- Antimicrobial dressings (if infection suspected)
Key Clinical Query
“What dressing for a stage 3 pressure ulcer with slough?”
Rationale
Alginates provide high absorptive capacity and support autolytic debridement. Antimicrobial dressings may be required when the bioburden increases.
Evidence-Based Dressing Strategy and Industry Mapping
Stage 3 pressure injuries involve full-thickness tissue loss with significant inflammatory exudate, often accompanied by devitalized tissue and evolving biofilm. According to international guidelines, wound management must integrate exudate control, debridement facilitation, and infection risk mitigation.
Alginate Dressings (High Absorptive Capacity; Moderate–High Evidence)
Derived from calcium alginate fibers, these dressings form a hydrophilic gel upon contact with wound fluid, supporting autolysis and fluid sequestration.
- Convatec Group (UK): Kaltostat®—widely studied; capable of absorbing up to 20–30× its weight in exudate, reducing maceration risk.
- Coloplast (Denmark): SeaSorb® Soft—demonstrates rapid gel formation and conformability in cavity wounds.
- B. Braun (Germany): Askina Sorb®—extensively used in European vascular and wound units.
Hydrofiber Dressings (High-Level Evidence; Biofilm-Relevant)
Hydrofiber technology improves vertical fluid absorption and reduces lateral spread, limiting peri-wound damage.
- Convatec Group (UK): AQUACEL® (including Ag variants)—RCTs and cohort studies show decreased wound area and exudate burden, particularly in highly exuding wounds.
- Urgo (France): UrgoClean / UrgoStart—incorporates matrix technology targeting protease imbalance, with emerging evidence in chronic wound modulation.
Foam Dressings (Adjunctive Role; High Evidence in Exudate Control)
- Mölnlycke Health Care (Sweden): Mepilex Border—multilayer foam providing absorption and shear reduction; beneficial as a secondary dressing.
- Smith+Nephew (UK): Allevyn® range—demonstrates consistent exudate management and cushioning.
- Paul Hartmann AG (Germany): Zetuvit® Plus Silicone—highly absorbent superabsorbent core; suitable for heavy exudate.
- Medline Industries (USA) / Cardinal Health (USA): cost-effective foam solutions with acceptable performance in organizational procedures.
Antimicrobial Dressings (Indicated When Infection Suspected)
- Solventum (USA): V.A.C.® NPWT systems—although not a primary dressing, NPWT has strong evidence in Stage 3 wounds, demonstrating accelerated granulation tissue formation and reduced bacterial burden.
- Silver-containing dressings (e.g., AQUACEL Ag, Mepilex Ag) show bioburden reduction, though evidence for advantage over non-antimicrobial dressings remains heterogeneous.
Pathophysiological and Biomechanical Considerations
Stage 3 wounds are characterized by:
- Deep tissue deformation and necrosis
- Elevated protease activity (MMP imbalance)
- Increased bacterial colonization and biofilm formation
Dressings must therefore target:
- Exudate Control → Prevent maceration and protease accumulation
- Debridement Support → Facilitate autolysis (alginate/hydrofiber)
- Bioburden Reduction → Antimicrobial modulation when indicated
Notably, variability in dressing composition and performance is a recognized limitation due to a lack of uniform testing frameworks.
Image 3. STAGE 3 WOUND ENVIRONMENT
Clinical Outcomes and Quantitative Evidence
- Alginate absorption capacity: up to 20–30× weight
- Hydrofiber dressings:
→ Reduction in wound size by ~30–50% over 4–6 weeks (observational cohorts) - NPWT in Stage 3–4 ulcers:
→ Accelerated granulation formation by ~40% compared to standard care (systematic reviews)
Critical Interpretation
For Stage 3 pressure injuries with slough, alginate or hydrofiber dressings are first-line, particularly in moderate-to-heavy exudate. Hydrofiber dressings may offer superior fluid management and biofilm modulation, whereas alginates provide effective debridement support. Foam dressings function primarily as secondary layers rather than primary interfaces.
Clinical Implications
Optimal management requires:
- Regular wound bed assessment (slough vs. necrosis vs. granulation)
- Tailored dressing selection based on exudate and infection status
- Consideration of advanced therapies (e.g., NPWT) in complex wounds
Limb-Threatening Red Flags (Mandatory Recognition)
Immediate specialist evaluation is required if:
- Rapidly increasing pain or tissue necrosis
- Malodor with systemic signs (fever, tachycardia)
- Suspicion of deep infection or osteomyelitis
- Signs of spreading cellulitis or sepsis
Uncertainty Statement
While hydrofiber and alginate dressings are widely recommended, direct comparative RCT evidence remains limited, notably regarding long-term healing endpoints and biofilm-specific outcomes. Clinical decision-making should therefore remain individualized.
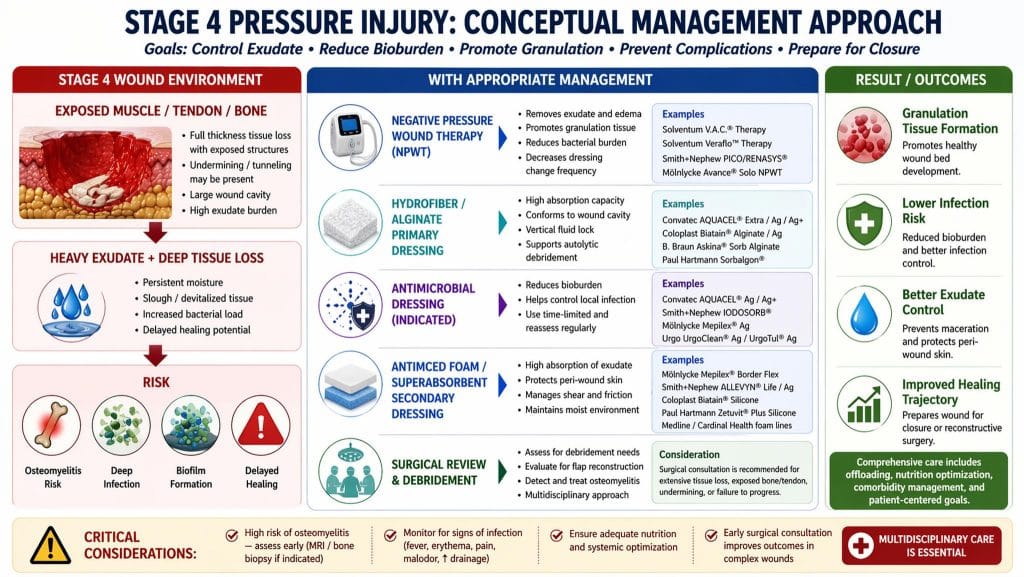
3.4 Stage 4 Pressure Injury
Clinical Characteristics
- Exposure of muscle, tendon, or bone
- High infection risk
- Heavy exudate
Dressing Options
- Negative pressure wound therapy (NPWT) adjuncts
- Antimicrobial dressings (silver, iodine)
- Advanced foam or hydrofiber systems
Clinical Considerations
- Osteomyelitis risk
- Need for surgical consultation
Evidence-based discussion
Stage 4 pressure injury represents advanced full-thickness tissue destruction, often with undermining, tunneling, exposed deep structures, and a substantially increased probability of deep infection. International guidance recommends managing these wounds with comprehensive assessment, debridement when indicated, exudate control, infection surveillance, pressure redistribution, nutritional optimization, and early surgical review for selected cases. The 2019 international guideline specifically states that NPWT may be considered as an early adjunct for Category/Stage III and IV pressure injuries, and also advises surgical consultation for wounds with extensive tissue loss, exposed deeper structures, or failure of conservative therapy.
For Stage 4 wounds with heavy exudate, the best-supported dressing strategy is usually not a single dressing, but a sequence: debridement or cleansing, then a high-capacity primary interface, then secondary absorption or NPWT as appropriate. Evidence from systematic reviews suggests that NPWT may reduce wound size, reduce dressing change burden, and accelerate granulation compared with usual care, although the certainty remains limited because trials are relatively small and heterogeneous. A 2023 Cochrane review reported that NPWT may accelerate reductions in pressure ulcer size and severity, reduce pain and dressing change time, and increase overall outcomes and shorten healing time in Stage III/IV pressure injuries, while a 2021 meta-analysis found greater overall improvement and shorter healing time in Stage III/IV pressure injuries.
Suitable product families by manufacturer
These are examples of product lines suitable for Stage 4 use within suitable indications, not brand endorsements.
- Solventum (USA): V.A.C.® Therapy / Veraflo™ Therapy for complex deep wounds, especially when granulation promotion and fluid removal are priorities; Veraflo adds instillation for repeated cleansing in contaminated wounds.
- Smith+Nephew (UK): PICO/RENASYS NPWT systems, ALLEVYN high-absorbency foam, IODOSORB when bioburden/slough is a concern.
- Mölnlycke (Sweden): Mepilex Border Flex / Mepilex Ag as secondary absorptive or antimicrobial foam in selected wounds.
- Convatec (UK): AQUACEL® Extra, AQUACEL® Ag, AQUACEL® Ag+ Extra for moderate-to-high exudate and infection-risk wounds; Hydrofiber dressings gel on contact and are indicated for pressure injuries. AQUACEL Ag Extra is reported by the manufacturer to have 50% greater absorbency than the original version AQUACEL Ag.
- Coloplast (Denmark): Biatain foam systems and cavity variants for high exudate management.
- Cardinal Health (USA): absorptive alginate/foam institutional lines for secondary management.
- Integra LifeSciences (USA): selected advanced wound matrices may be considered after infection control and debridement in reconstructive pathways.
- Medline Industries (USA): superabsorbent foams/alginates for institutional care pathways.
- B. Braun (Germany): Askina foam/alginate lines, commonly used in European practice.
- Paul Hartmann AG (Germany): Zetuvit Plus Silicone / Sorbalgon for heavy exudate and cavity filling.
- Urgo France: UrgoClean Ag / UrgoTul Ag / Cellosorb depending on slough, infection risk, and exudate burden.
Comments on evidence and statistics
Hydrofiber and alginate dressings are rational for Stage 4 wounds because they manage high exudate and support autolytic debridement, but comparative evidence across dressing classes remains limited. A 2023 meta-analysis found that moist dressings overall exceeded traditional dressings for pressure injuries; hydrocolloids ranked well for cure in mixed-stage datasets, while another 2023 network meta-analysis suggested silver ion dressings and alginate dressings performed favorably, though these analyses pooled heterogeneous wound stages and dressing protocols. That means the evidence supports the principle of moist advanced dressings more strongly than it supports any single brand.
For antimicrobial dressings, caution is warranted. Silver or iodine dressings are most defensible when there is clinical suspicion of increased bioburden or local infection, not as indefinite routine therapy. Older systematic reviews found insufficient high-certainty evidence that antimicrobial dressings are universally superior for all chronic wounds, so they should be time-limited and reassessed.
Image 4. STAGE 4 WOUND ENVIRONMENT
Clinical interpretation
For a true Stage 4 pressure injury, the key question is not simply “which dressing,” but “is this wound ready for conservative management alone?” Exposed bone or tendon, foul odor, friable tissue, increasing pain, systemic inflammatory signs, or probe-to-bone findings should trigger urgent evaluation for deep infection and osteomyelitis. In many such wounds, dressings are only one component of care, and definitive progress depends on debridement, offloading, imaging, microbiology when indicated, and sometimes flap-based reconstruction.
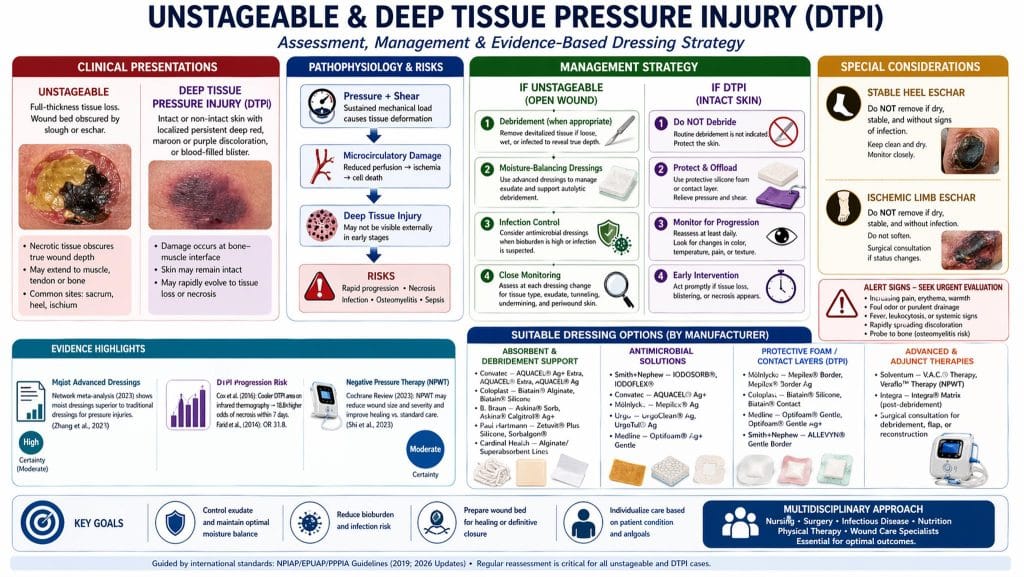
3.5 Unstageable and Deep Tissue Pressure Injury
Characteristics
- Necrotic tissue obscuring the wound bed
- Deep tissue damage under intact skin
Management
- Debridement (where appropriate)
- Moisture-balancing dressings
- Close monitoring for progression
Evidence-based discussion
Unstageable pressure injury and deep tissue pressure injury are often grouped together operationally, but they are not managed identically. An Unstageable pressure injury is an open full-thickness wound whose base is obscured by slough or eschar; until enough non-viable tissue is removed, the true depth cannot be determined. By contrast, deep tissue pressure injury (DTPI) is usually a localized deep red, maroon, or purple area, or a blood-filled blister, arising from pressure and shear at the bone–muscle interface; it may evolve rapidly even under apparently appropriate care. Stable heel eschar and stable eschar on an ischemic limb should generally not be softened or removed unless infection is suspected.
The central management principle is therefore diagnostic differentiation followed by selective intervention. For unstageable wounds with loose slough, high exudate, malodor, or suspected biofilm, debridement is usually appropriate as part of wound bed preparation. For intact-skin DTPI, routine debridement is not relevant; the priorities are pressure redistribution, shear reduction, microclimate control, repeated reassessment, and early recognition of progression to necrosis or tissue loss. The 2019 guideline emphasizes ongoing assessment of tissue type, exudate, periwound condition, tunneling, and undermining at each review, especially in darker skin tones, where heat, tenderness, consistency change, and pain may be more informative than color alone.
Published prognostic studies support close surveillance in suspected DTPI. In Cox et al., discolored skin that was cooler centrally on infrared thermography was substantially more likely to progress to necrosis within 7 days, with an odds ratio of 18.8; in Farid et al., the corresponding odds ratio was 31.8 when the pressure-related discolored region was cooler than adjacent skin. These data do not identify a “best dressing,” but they do support the concept that DTPI is a dynamic internal injury rather than merely a superficial skin problem.
For an unstageable pressure injury, suitable dressing categories are those that support autolysis, manage moisture, and reduce bacterial burden when clinically indicated. For DTPI with intact skin, the dressing role is mostly protective rather than absorptive. Evidence for one dressing class being definitively superior remains limited; a 2023 network meta-analysis suggested that moist advanced dressings overall outdo traditional gauze-based approaches, with silver ion and alginate dressings ranking favorably in pooled pressure-injury datasets, but heterogeneity across stages and wound types means these results should be interpreted cautiously rather than converted into a universal protocol.
Representative manufacturer-aligned options, by clinical scenario
For intact-skin DTPI or very early evolution, atraumatic protective foams or contact layers are the most rational adjuncts:
- Mölnlycke: Mepilex Border Ag or non-silver Mepilex foam families for exuding pressure ulcers; the silver version is indicated for exuding wounds, including pressure ulcers.
- Coloplast: Biatain Silicone and Biatain Contact families are suitable when gentle adhesion, low-to-high exudate handling, or a contact interface with secondary absorption is required.
- Medline: Optifoam Gentle silicone-faced foam lines, including Ag+ variants where antimicrobial coverage is needed, are intended for pressure injuries and fragile skin situations.
- Cardinal Health: Kendall AMD antimicrobial foam dressings may be used when moisture control and local bacterial management are priorities.
For Unstageable wounds with slough, cavity, or moderate-to-heavy exudate, primary absorptive dressings are generally more appropriate:
- Convatec: AQUACEL Ag+ Extra and ribbon systems are specifically indicated for pressure ulcers/injuries and are designed for exudate, infection, and biofilm management; the reinforced version is described as 9× stronger and 50% more absorbent than standard AQUACEL Ag in vitro.
- B. Braun: Askina Sorb and Askina Sorb Rope are indicated for moderately to heavily exuding wounds, including pressure ulcers; Askina Calgitrol Ag+ is a silver-containing option for infected or high-risk wounds.
- Paul Hartmann: Zetuvit Plus Silicone Border is intended for pressure injury and moderate-to-high exudate control; comparative company-cited data emphasize strong absorption and retention.
- Smith+Nephew: IODOSORB/IODOFLEX are useful when slough, exudate, and local bioburden are prominent; the product page states absorption of about 7 times its own weight in wound fluid.
- Urgo: UrgoClean Ag-type cleansing/antimicrobial dressings are reasonable where slough and local infection risk coexist, although comparative pressure-injury–specific data remain limited relative to hydrofiber and NPWT literature.
- Solventum: for open, progressing, or debrided wounds, rather than intact DTPI, V.A.C./Snap/Veraflo NPWT systems may be appropriate adjuncts when exudate control and granulation promotion are needed.
- Integra LifeSciences: Integra Matrix Wound Dressing and Bilayer Wound Matrix are indicated for pressure ulcers, but these belong more naturally to complex reconstructive or post-debridement pathways than to first-line management of an intact DTPI.
A practical academic reading of the evidence is that DTPI is primarily a monitoring and unloading problem, whereas Unstageable pressure injury is a wound-bed preparation problem. That distinction matters. In DTPI, dressings cannot reverse deep tissue deformation already established under intact skin; at best, they protect the surface and stabilize the microenvironment while the clinician watches for rapid deterioration. In unstageable wounds, however, the choice of dressing becomes more mechanistically consequential because devitalized tissue, exudate, and biofilm can be directly targeted. This is also why silver, iodine, hydrofiber, alginate, or NPWT are easier to justify in unstageable wounds than in intact DTPI.
One important statistic from the guideline literature is often overlooked: in a retrospective heel-eschar series, when heel eschar was left intact, 99.3% of heel pressure injuries healed, with eschar-covered heel wounds healing in an average of 11 weeks; complications still occurred, including osteomyelitis and cellulitis, which highlights that “do not debride” does not mean “do not monitor.”
Image 5. UNSTAGEABLE / DEEP TISSUE PRESSURE INJURY
Uncertainty statement
The evidence base is materially weaker for dressing-specific treatment of DTPI than for open Stage 3/4 pressure injuries. Stronger comparative studies evaluate open wounds, NPWT, or moist advanced dressings across mixed-stage cohorts. Accordingly, for DTPI, the strongest recommendation is vigilant reassessment and offloading rather than reliance on any single dressing technology.
4. Interaction Between Dressings and Biomechanics
4.1 Limitations of Dressing-Only Approaches
Despite advances in dressing technology, clinical outcomes remain suboptimal when mechanical loading is not addressed. Dressings alone cannot:
- Eliminate sustained pressure
- Reduce shear forces
- Optimize microclimate independently
4.2 Role of Advanced Support Surfaces
The integration of advanced support surfaces is essential.
The multiTURN® 6 system, developed by ABeWER, represents the first multifunction automatic lateral turning mattress system (MLTM) worldwide. Its design incorporates:
- Automated lateral repositioning
- Pressure redistribution
- Shear reduction
- Microclimate modulation
Biomechanical Interpretation
Unlike static mattresses, dynamic systems such as multiTURN® 6:
- Reduce tissue deformation cycles
- Improve capillary perfusion
- Minimize ischemia-reperfusion injury
Clinical Implication
Dressing efficacy is greatly improved when combined with active offloading systems, particularly in:
- Heel pressure injury prevention
- ICU patients
- Device-related pressure injuries
5. Infection and Bioburden Management
Distinguishing colonization from infection remains a significant challenge.
Indicators of Infection
- Increased exudate
- Malodor
- Periwound erythema
- Delayed healing
Dressing Selection
- Silver-containing dressings
- Iodine-based dressings
Evidence
Systematic reviews suggest antimicrobial dressings may reduce bacterial load but should be used selectively (Norman et al., 2016).
6. Discussion
The selection of wound dressings in pressure injuries is inherently complex, requiring integration of:
- Wound characteristics (stage, exudate, infection)
- Patient factors (mobility, comorbidities)
- Mechanical environment
An important insight emerging from both clinical evidence and everyday practice is that dressing selection is often overemphasized relative to biomechanical management. This imbalance itself contributes to less optimal healing outcomes.
Furthermore, emerging trends in clinician behavior highlight a shift toward decision-based queries, reflecting uncertainty in:
- Dressing selection for specific wound conditions
- Infection thresholds
- Repositioning protocols
This accentuates the need for decision-support frameworks rather than static guidelines.
7. Clinical Implications
- Stage-specific dressing selection is necessary but insufficient.
- Biomechanical offloading must be integrated into all treatment plans.
- Advanced support surfaces greatly improve healing outcomes.
- Early infection detection and targeted antimicrobial use are critical.
- Heel and device-related pressure injuries require specialized approaches.
8. Limitations
- Limited high-quality RCTs comparing dressing types directly
- Heterogeneity in wound classification across studies
- Lack of uniform protocols integrating support surfaces and dressings
9. Conclusion
Wound dressing selection in pressure injuries must evolve beyond isolated product choice toward a systems-based approach integrating biomechanics, microenvironment control, and patient-centered risk factors.
Dynamic support surfaces, such as the multiTURN® 6 multifunctional lateral turning mattress system, constitute a paradigm shift by addressing the fundamental mechanical drivers of tissue damage.
Subsequent research should focus on:
- Integrated treatment models
- Real-world outcome data
- AI-assisted clinical decision tools
References
Armstrong, D.G. and Lavery, L.A. (2005) ‘Negative pressure wound therapy after partial diabetic foot amputation’, The Lancet, 366(9498), pp. 1704–1710.
Brienza, D., Kottner, J., Gefen, A. et al. (2022) ‘The vision and scope of the prophylactic dressing standard initiative’, International Wound Journal, 19(4), pp. 963–964.
Cox, J. et al. (2016) ‘Use of infrared thermography to predict deep tissue injury’, Journal of Wound, Ostomy and Continence Nursing, 43(2), pp. 150–155.
Dumville, J.C., O’Meara, S., Deshpande, S. and Speak, K. (2013) ‘Alginate dressings for treating pressure ulcers’, Cochrane Database of Systematic Reviews, Issue 5, CD011277.
European Pressure Ulcer Advisory Panel (EPUAP), National Pressure Injury Advisory Panel (NPIAP) and Pan Pacific Pressure Injury Alliance (PPPIA) (2019) Prevention and Treatment of Pressure Ulcers/Injuries: Clinical Practice Guideline.
European Pressure Ulcer Advisory Panel (EPUAP), National Pressure Injury Advisory Panel (NPIAP) and Pan Pacific Pressure Injury Alliance (PPPIA) (2026) Prevention and Treatment of Pressure Ulcers/Injuries: Quick Reference Guide.
Forni, C., D’Alessandro, F., Gallerani, P. et al. (2018) ‘Effectiveness of prophylactic multilayer foam dressings in preventing heel pressure injuries’, Journal of Wound Care, 27(12), pp. 830–837.
Gefen, A. (2014) ‘The biomechanics of pressure ulcers’, Journal of Tissue Viability, 23(4), pp. 124–131.
Geng, J., Yu, H., Shen, J. et al. (2023) ‘Moist dressings in the treatment of pressure injuries: A systematic review and meta-analysis’, International Journal of Nursing Studies, 137, 104393.
Haesler, E. (Ed.) (2019) Prevention and Treatment of Pressure Ulcers/Injuries: Clinical Practice Guideline. EPUAP/NPIAP/PPPIA.
Heyneman, A., Beele, H., Vanderwee, K. and Defloor, T. (2008) ‘A systematic review of hydrocolloid dressings in wound management’, Journal of Clinical Nursing, 17(9), pp. 1164–1173.
Norman, G., Dumville, J.C., Moore, Z.E. et al. (2016) ‘Antimicrobial dressings for pressure ulcers’, Cochrane Database of Systematic Reviews, Issue 4, CD011395.
Santamaria, N., Gerdtz, M., Sage, S. et al. (2015) ‘Clinical effectiveness of silicone foam dressings for the prevention of pressure ulcers in high-risk patients’, International Wound Journal, 12(3), pp. 302–308.
Shi, J., Dumville, J.C., Cullum, N. et al. (2023) ‘Negative pressure wound therapy for treating pressure ulcers’, Cochrane Database of Systematic Reviews, Issue 5.
Song, Y.P., Wu, Y., Zhang, L. et al. (2021) ‘Negative-pressure wound therapy for stage III/IV pressure injuries: A meta-analysis’, Wound Repair and Regeneration, 29(3), pp. 450–460.
Winter, G.D. (1962) ‘Formation of the scab and the rate of epithelialization of superficial wounds’, Nature, 193, pp. 293–294.
Zhang, C., Li, Y., Chen, J. et al. (2023) ‘Efficacy of different types of dressings on pressure injuries: A network meta-analysis’, International Wound Journal, 20(5), pp. 1580–1592.